30-Day Pilot Evaluation
Intra-Articular Joint Fluid Replacement Medical Device

Sponsor: Novanto Technologies, LLC | Investigating Veterinarian: Dr. Hadley Whitting, DVM

Population:
9
Sterile Saline Controls
30
Days Duration
9
Condranol
Subjects
11
Horses
1. Background
Horses with performance-related joint pain and lameness are commonly managed with intra-articular therapies aimed at improving joint comfort and functional movement. Condranol is a newly launched intra-articular joint fluid replacement medical device intended to support joint function.
2. Objective
To evaluate short-term (30-day) changes in limb-level lameness grade following intra-articular administration of Condranol compared with sterile saline control injections in horses exhibiting baseline lameness.
3. Study Design
Controlled, 30-day pilot evaluation in horses with baseline limb lameness. Horses were assigned to either Condranol (n=9) or sterile saline control (n=2).
4. Treatments
Condranol group (n=9): Intra-articular Condranol injections were administered into one or more joints within the affected limb. Treating multiple joints within a single affected limb is consistent with common sports medicine practice when clinical signs indicate multi-joint contribution within that limb. Total injected volume was recorded.
Control group (n=2): Intra-articular sterile saline injections (sham comparator) were administered similarly.
5. Lameness Evaluation Method
Scoring system: AAEP lameness scale (0–5), with 0.5 grade increments permitted.
Assessment conditions: Trot on a circle on the same surface at Day 0 and Day 30.
Endpoint recorded: Limb-level lameness grade for the affected limb
(even when multiple joints in that limb were treated).
6. Endpoints
Primary endpoint: Change in AAEP lameness grade from Day 0 to Day 30 for the affected limb.
Secondary observations: Joint(s) treated and total injected volume per horse/limb were recorded.
Additional clinical fields (effusion, heat, pain on palpation, flexion response) were not consistently
available in this initial dataset.
7. Results Summary
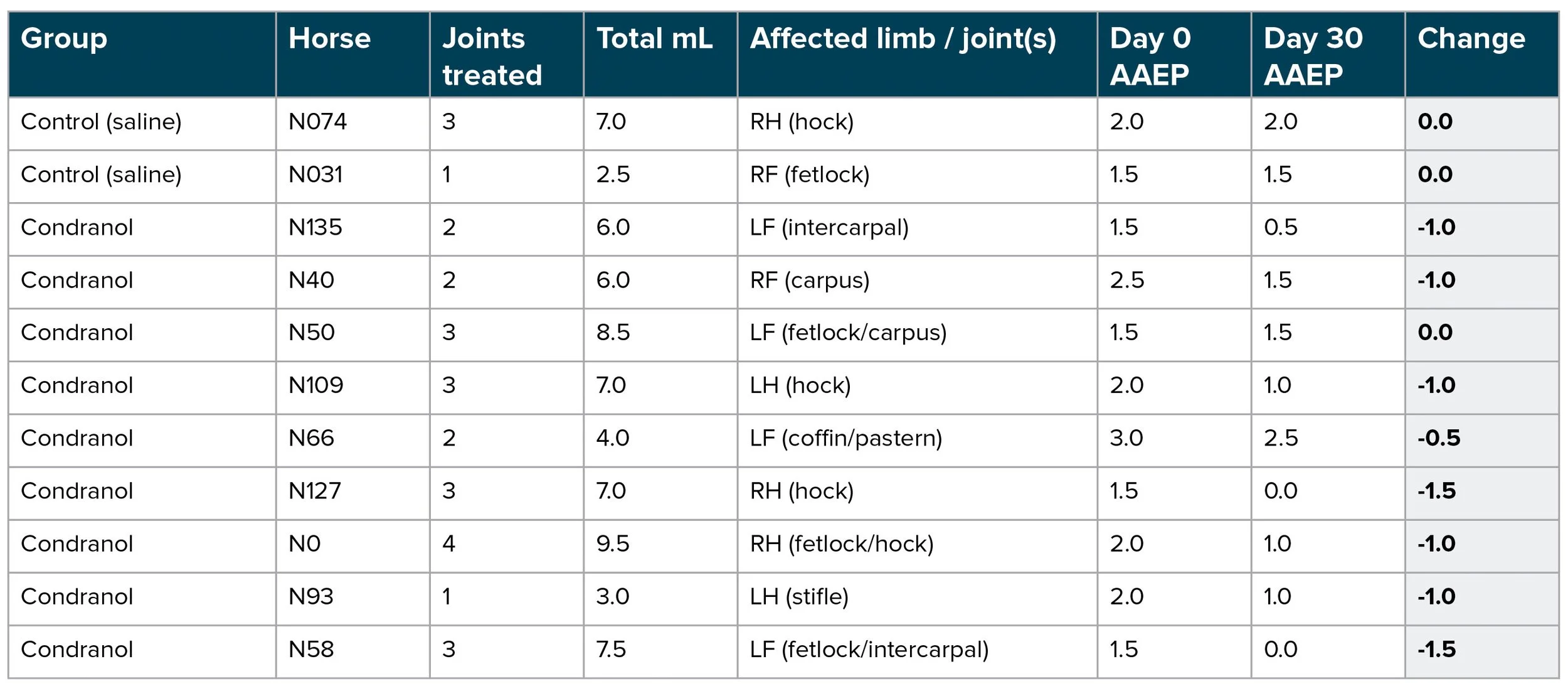
Controls (n=2): 0/2 improved; 2/2 had no change; 0/2 worsened.
Condranol subjects (n=9): 8/9 improved by at least 0.5 grade; 7/9 improved by at least 1.0 grade; 0/9 worsened.
Condranol Subjects:
7/9
improved by at least 1.0 grade
8/9
improved by at least 0.5 grade
0/9
worsened
Mean AAEP lameness grade (Condranol group): 1.94 at Day 0 to 1.00 at Day 30.
Mean change: -0.94 grades (improvement); median change: -1.0 grade.
8. Horse-Level Outcome Table (Day 0 vs Day 30)
9. Interpretation
In this 30-day controlled pilot evaluation, 8 out of 9 Condranol-treated horses demonstrated improvement in limb-level AAEP lameness grade by Day 30, while the two sterile saline control horses showed no change. These findings support further evaluation in a larger study with additional standardized clinical findings and blinded scoring.
10. Limitations
Small sample size, particularly the control group (n=2).
Pilot design; this dataset does not include consistent standardized secondary clinical fields
(effusion, heat, pain, flexion response).
Lameness grading in this initial phase was not designed as a blinded assessment.

